Research
The human brain contains roughly 100 billion (1011) neurons and 100 trillion (1014) synaptic connections, with tremendous biochemical complexity at each synapse. Synapses are not simple on/off switches, conducting signals from one neuron to another. They can be thought of as tunable switches; with subtle modifications in the strengths of these synaptic communications being central to higher brain functions.
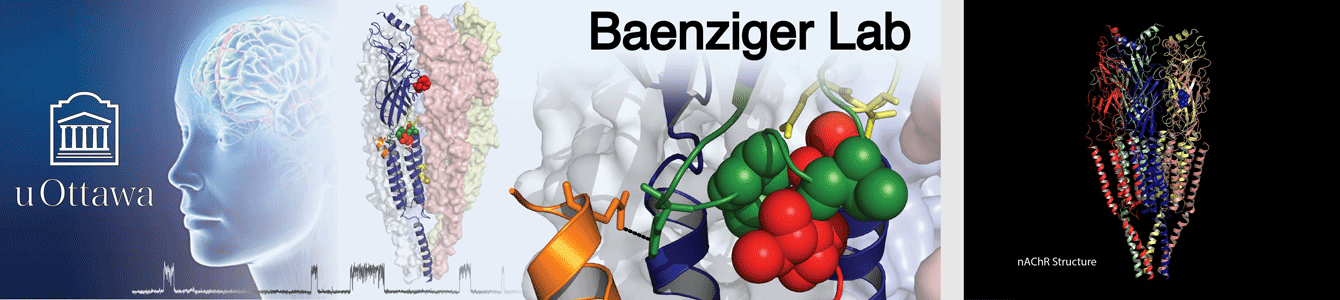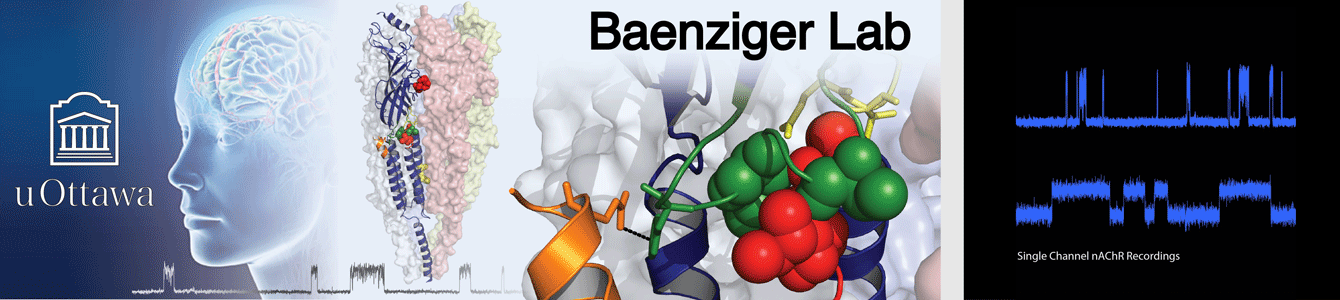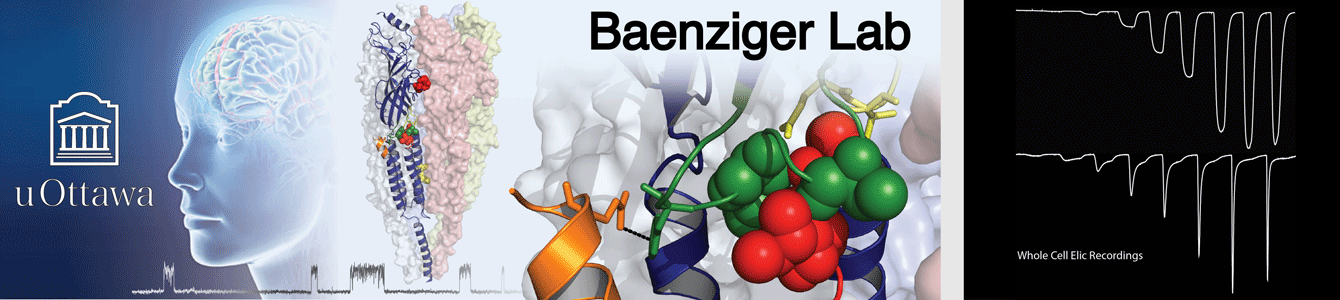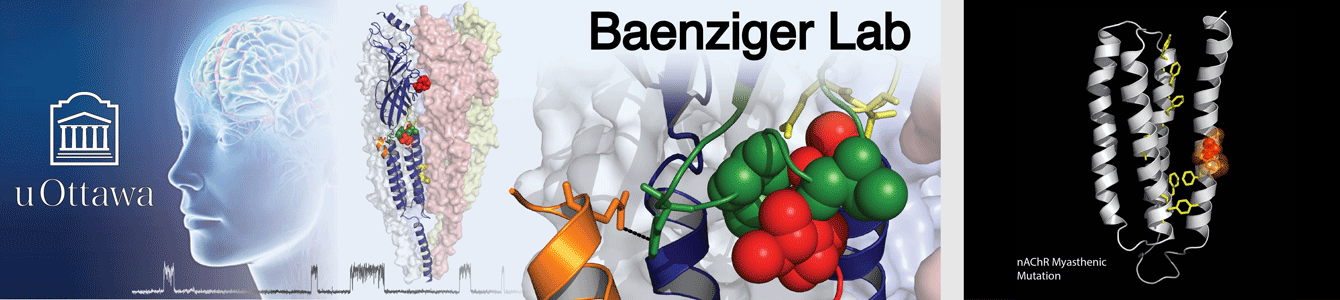
We study the structure and ultimately function of a super-family of proteins, called pentameric ligand-gated ion channels (pLGICs), that play a central role in synaptic communication. This super-family includes fast-acting neurotransmitter receptors/ion channels that respond to acetylcholine, glycine, GABA, and 5-hydroxytryptamine (serotonin), etc. These receptors/ion channels are implicated in neurological processes/diseases and are pharmaceutical targets. Recent structural information is beginning to shed light on the molecular basis of both pLGIC function and pLGIC-drug interactions.
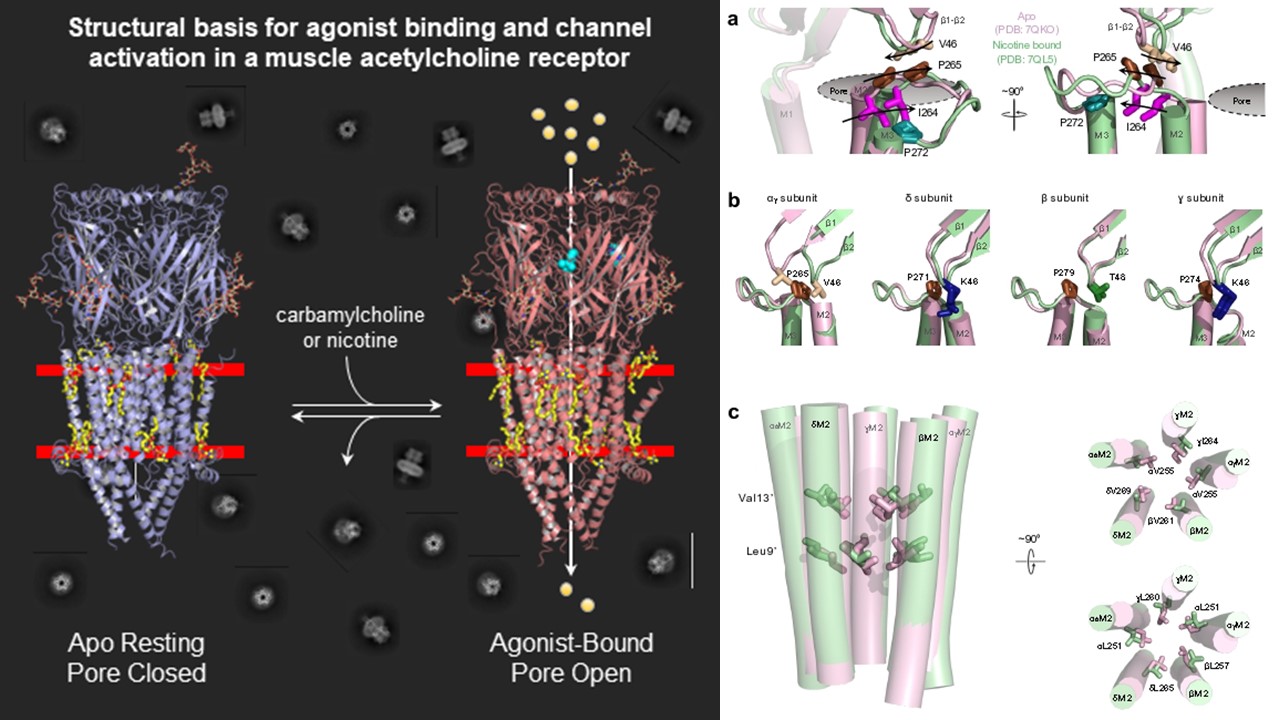
Left: Zarkadas et al., Conformational transitions and ligand-binding to a muscle-type nicotinic acetylcholine receptor. Neuron (2022) 110, 1358-1370
Right: Thompson et al., A release of local subunit conformational heterogeneity underlies gating in a muscle nicotinic acetylcholine receptor. Nat. Commum. (2024) 15 (1), 1803, DOI: 10.1038/s41467-024-46028-x
Most of the research in our lab focuses on the muscle-type nicotinic acetylcholine receptor (nAChR). We use cryo-electron microscopy to understand the structural basis of nAChR function, including how nAChR gating is influenced by agonists, toxins, pharmaceutical products, etc. Fundamental knowledge of how the nAChR and other pLGICs respond to allosteric modulators is essential for understanding the mechanisms underlying the altered synaptic communication that occurs over the course of human disease. We are particularly interested in understanding how lipids modulate function of the nAChR and other pLGICs.
Our structural studies are complemented by electrophysiological measurements, molecular dynamics (MD) simulations, and other biochemical/biophysical approaches. Research in the Baenziger lab is supported and/or driven by collaborations with outstanding scientists, such as Drs. Hugues Nury and Eleftherios Zarkadas (IBS, Grenoble, France), François Dehez (CNRS , University of Lorraine, Nancy, France), Chris Ulens (KU Leuven, Leuven, Belgium), and Corrie daCosta (University of Ottawa, Ottawa, Canada). The Baenziger and daCosta labs hold a joint monthly ion channel journal club.